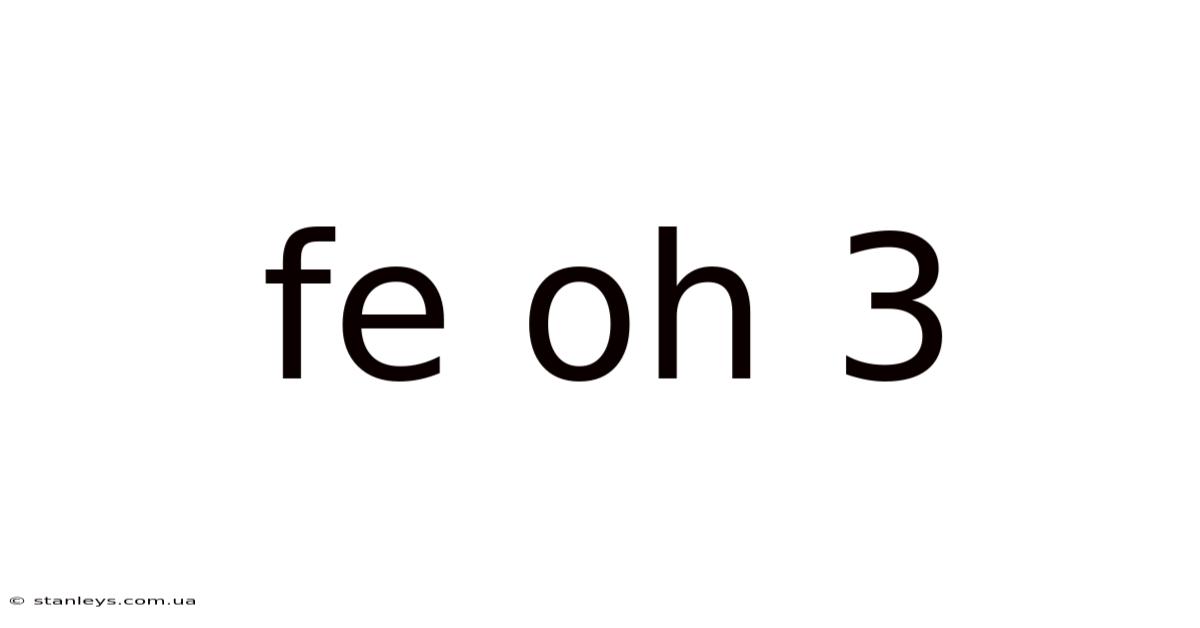Unveiling the Mysteries of Fe(OH)₃: Properties, Synthesis, and Applications of Iron(III) Hydroxide
Iron(III) hydroxide, often represented as Fe(OH)₃, is a fascinating compound with a rich history and a wide range of applications. This article breaks down the intriguing world of Fe(OH)₃, exploring its properties, various synthesis methods, and its significant roles across diverse industries. But understanding Fe(OH)₃ is crucial not only for chemistry students but also for professionals in fields like materials science, environmental engineering, and water treatment. We will explore its unique characteristics, break down the intricacies of its synthesis, and highlight its importance in various applications.
Introduction: What is Fe(OH)₃?
Fe(OH)₃, also known as ferric hydroxide, is an inorganic compound composed of iron(III) cations (Fe³⁺) and hydroxide anions (OH⁻). Still, it doesn't exist as a simple molecule but rather forms a complex structure, often hydrated and poorly defined, typically appearing as a reddish-brown precipitate. This seemingly simple compound exhibits a surprising complexity in its behaviour and properties, making it a subject of ongoing scientific interest. Its unique characteristics stem from the variable oxidation state of iron and its ability to form different crystalline structures and hydrates, depending on the conditions of its synthesis. The understanding of these nuances is critical to controlling its properties for specific applications And that's really what it comes down to..
Properties of Fe(OH)₃: A Closer Look
Fe(OH)₃ possesses a number of key properties that define its behaviour and applications. These include:
- Appearance: Typically a reddish-brown gelatinous precipitate, though its precise appearance can vary depending on the preparation method and degree of hydration.
- Solubility: Fe(OH)₃ is virtually insoluble in water, a crucial property in many of its applications. Its solubility is however influenced by pH; it's more soluble in acidic solutions due to the formation of soluble iron(III) complexes.
- Amphoteric Nature: While predominantly acting as a base, Fe(OH)₃ exhibits amphoteric behaviour, meaning it can react with both acids and bases. It dissolves in strong acids to form iron(III) salts and reacts with strong bases to form ferrites.
- Magnetic Properties: Fe(OH)₃ displays weak paramagnetic properties due to the presence of unpaired electrons in the iron(III) ion. This property is utilized in certain applications.
- Reactivity: Fe(OH)₃ is a relatively reactive compound and readily participates in redox reactions, acting as both an oxidizing and reducing agent depending on the reaction conditions. It can also undergo dehydration to form iron(III) oxide (Fe₂O₃).
Synthesis of Fe(OH)₃: Different Approaches
Several methods exist for synthesizing Fe(OH)₃, each yielding slightly different characteristics in the final product. The choice of method depends on the desired properties and intended application. The most common methods include:
-
Precipitation from Aqueous Solution: This is the most common method. Aqueous solutions of iron(III) salts, such as iron(III) chloride (FeCl₃) or iron(III) nitrate (Fe(NO₃)₃), are reacted with a base, typically sodium hydroxide (NaOH) or ammonium hydroxide (NH₄OH). The reaction produces Fe(OH)₃ as a precipitate:
Fe³⁺(aq) + 3OH⁻(aq) → Fe(OH)₃(s)
The reaction conditions, such as temperature, concentration, and pH, significantly influence the particle size, morphology, and crystallinity of the resulting Fe(OH)₃ Not complicated — just consistent..
-
Hydrothermal Synthesis: This method involves reacting iron(III) salts with a base in an autoclave at elevated temperatures and pressures. Hydrothermal synthesis allows for better control over the particle size and morphology of the Fe(OH)₃ product. It’s particularly useful for obtaining well-defined crystalline structures Not complicated — just consistent. And it works..
-
Sol-Gel Method: The sol-gel process involves the hydrolysis and condensation of iron(III) alkoxides, yielding Fe(OH)₃ as a gel. This method offers excellent control over the porosity and surface area of the final product, making it suitable for applications requiring high surface area materials Which is the point..
Dehydration to Iron(III) Oxide (Fe₂O₃): A Crucial Transformation
Upon heating, Fe(OH)₃ readily undergoes dehydration to form iron(III) oxide (Fe₂O₃), also known as ferric oxide or hematite:
2Fe(OH)₃(s) → Fe₂O₃(s) + 3H₂O(g)
This dehydration process is often employed to obtain Fe₂O₃, a crucial material in various industrial applications. The temperature and atmosphere during dehydration significantly influence the crystalline structure and properties of the resulting Fe₂O₃.
Applications of Fe(OH)₃: A Multifaceted Compound
The unique properties of Fe(OH)₃ translate into a wide range of applications across several industries. Some key applications include:
-
Water Treatment: Fe(OH)₃'s insolubility and ability to adsorb impurities make it an effective coagulant and flocculant in water treatment. It helps remove suspended solids, organic matter, and other contaminants from water, resulting in cleaner and safer drinking water.
-
Wastewater Treatment: Similar to its role in water treatment, Fe(OH)₃ finds application in treating industrial wastewater. It can remove heavy metals, phosphates, and other pollutants from wastewater before discharge, protecting the environment.
-
Pigment Production: The reddish-brown color of Fe(OH)₃ makes it a valuable pigment in various applications, although it's often converted to Fe₂O₃ for better stability. It's used in paints, cosmetics, and other products requiring color.
-
Catalysis: Fe(OH)₃ acts as a catalyst or catalyst precursor in several chemical reactions. Its catalytic activity stems from its redox properties and the ability to form various surface complexes It's one of those things that adds up. Nothing fancy..
-
Electrochemical Applications: Fe(OH)₃'s redox properties have made it attractive for electrochemical applications, including battery materials and electrocatalysis. Research explores its potential in various energy storage and conversion technologies.
-
Synthesis of Other Iron Compounds: Fe(OH)₃ serves as a precursor for the synthesis of other iron compounds, such as iron oxides, iron oxyhydroxides, and ferrites. This makes it a crucial intermediate in various materials synthesis processes That's the part that actually makes a difference..
Frequently Asked Questions (FAQs)
Q: Is Fe(OH)₃ toxic?
A: While Fe(OH)₃ itself is generally considered non-toxic, excessive exposure can cause irritation. That said, the toxicity depends on the form and purity of the compound and the route of exposure. Proper handling and safety precautions are always advised And it works..
Q: What is the difference between Fe(OH)₃ and Fe₂O₃?
A: Fe(OH)₃ is iron(III) hydroxide, containing hydroxide ions (OH⁻). Fe₂O₃ is iron(III) oxide, lacking hydroxide ions. Fe(OH)₃ dehydrates to form Fe₂O₃ upon heating.
Q: How is the purity of Fe(OH)₃ determined?
A: The purity of Fe(OH)₃ can be determined through various analytical techniques, including gravimetric analysis, titration, and instrumental methods like X-ray diffraction (XRD) and thermogravimetric analysis (TGA).
Q: What are the environmental concerns associated with Fe(OH)₃?
A: While Fe(OH)₃ itself is not considered a major environmental pollutant, improper disposal can lead to water contamination or soil changes. Its use in wastewater treatment, however, aims to mitigate environmental problems caused by pollutants Worth knowing..
Q: What are the future prospects for Fe(OH)₃ research?
A: Research on Fe(OH)₃ continues to focus on understanding its structure-property relationships, developing novel synthesis methods for tailored properties, and exploring its potential in advanced applications such as energy storage, catalysis, and environmental remediation Most people skip this — try not to..
Conclusion: A Versatile Compound with Broad Applications
Fe(OH)₃, despite its seemingly simple chemical formula, is a remarkable compound with diverse properties and widespread applications. Its versatility stems from its amphoteric nature, its ability to undergo dehydration, and its interaction with various substances. This leads to from its crucial role in water purification to its potential in advanced technologies, Fe(OH)₃ continues to be a subject of intense research and development, promising innovative solutions in various fields. Understanding its properties and synthesis methods is key to harnessing its full potential for the benefit of society and the environment. Further research will undoubtedly uncover even more fascinating aspects of this ubiquitous and important compound Surprisingly effective..