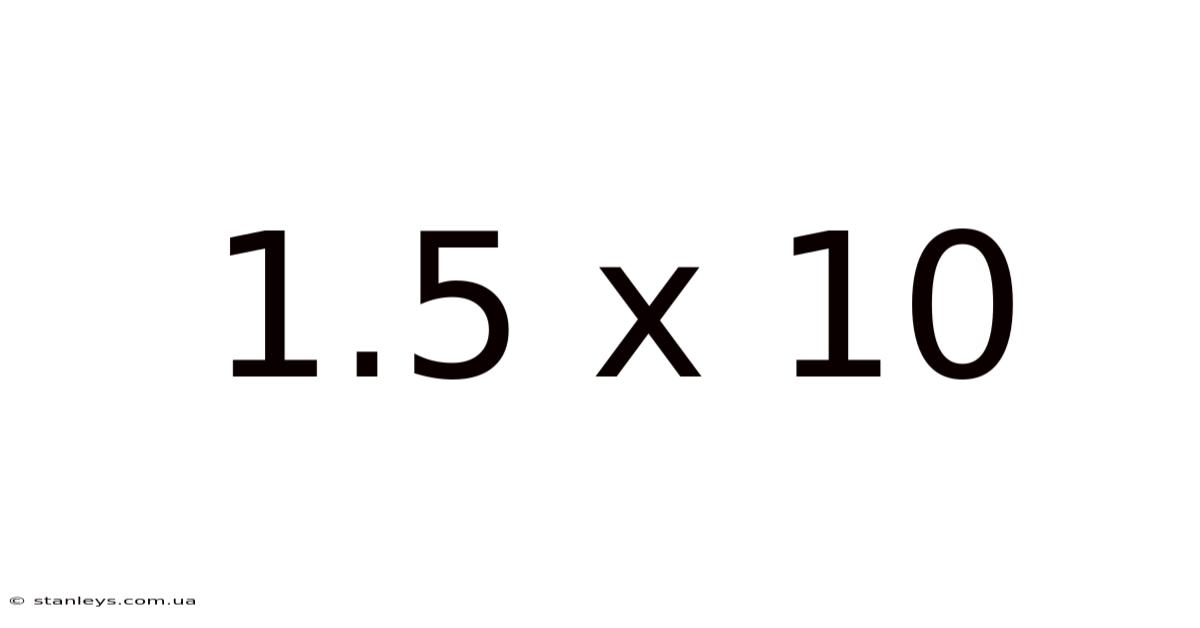Decoding 1.5 x 10: Exploring Scientific Notation and its Applications
This article looks at the seemingly simple expression "1.5 x 10," revealing its significance within the realm of scientific notation and its widespread applications across various scientific disciplines. We'll explore what scientific notation is, why it's crucial, how to work with it, and examine its relevance in real-world scenarios, from understanding astronomical distances to comprehending the microscopic world. This full breakdown will demystify this fundamental concept, making it accessible to everyone, regardless of their prior mathematical background That's the whole idea..
Understanding Scientific Notation
Scientific notation, also known as standard form, is a standardized way of writing very large or very small numbers. On top of that, it's based on expressing numbers as a product of a coefficient (a number between 1 and 10) and a power of 10. The general format is: a x 10<sup>b</sup>, where 'a' is the coefficient and 'b' is the exponent.
The expression "1.On top of that, 5 should be moved one place to the right. 5 x 10<sup>1</sup>**, or simply 15. And the exponent '1' indicates that the decimal point in 1. 5 x 10" itself is a simplified form of scientific notation. Still, while technically correct, it’s more commonly seen expanded as **1. Understanding the significance of the exponent is key to mastering scientific notation No workaround needed..
Why Use Scientific Notation?
Scientific notation offers several crucial advantages:
- Conciseness: It provides a compact way to represent extremely large or small numbers. Imagine trying to write out the number of atoms in a gram of gold without scientific notation; it would be incredibly cumbersome.
- Clarity: It improves the readability of numbers, especially those with numerous digits. This clarity is essential for avoiding errors and facilitating easy comprehension.
- Standardization: It creates a consistent format for presenting numerical data, promoting effective communication among scientists and researchers.
- Ease of Calculation: Scientific notation simplifies calculations involving very large or small numbers, making them significantly easier to perform.
Working with Scientific Notation: Examples and Calculations
Let's explore how to work with numbers expressed in scientific notation, focusing on both multiplication and division And that's really what it comes down to. Worth knowing..
Multiplication: To multiply two numbers in scientific notation, multiply their coefficients and add their exponents.
- Example: (2.5 x 10<sup>3</sup>) x (3 x 10<sup>2</sup>) = (2.5 x 3) x 10<sup>(3+2)</sup> = 7.5 x 10<sup>5</sup>
Division: To divide two numbers in scientific notation, divide their coefficients and subtract their exponents.
- Example: (6 x 10<sup>6</sup>) / (2 x 10<sup>2</sup>) = (6/2) x 10<sup>(6-2)</sup> = 3 x 10<sup>4</sup>
Addition and Subtraction: Adding or subtracting numbers in scientific notation requires a little more care. You must first confirm that both numbers have the same exponent. Then, add or subtract the coefficients, keeping the same exponent.
-
Example: (2.5 x 10<sup>3</sup>) + (1.5 x 10<sup>3</sup>) = (2.5 + 1.5) x 10<sup>3</sup> = 4 x 10<sup>3</sup>
-
Example (with adjustment): (2.5 x 10<sup>3</sup>) + (1.5 x 10<sup>2</sup>) = (2.5 x 10<sup>3</sup>) + (0.15 x 10<sup>3</sup>) = 2.65 x 10<sup>3</sup>
Real-World Applications of Scientific Notation
The applications of scientific notation are vast and span numerous fields:
-
Astronomy: Distances in space are often expressed using scientific notation. Take this case: the distance from Earth to the Sun is approximately 1.5 x 10<sup>8</sup> kilometers. Describing this distance without scientific notation would involve a long string of digits, making it difficult to manage and interpret.
-
Physics: In physics, especially at the atomic and subatomic levels, we encounter extremely small numbers, often representing measurements like the size of atoms or the mass of electrons. Scientific notation simplifies these representations and makes calculations more manageable. Here's one way to look at it: the mass of an electron is approximately 9.1 x 10<sup>-31</sup> kilograms.
-
Chemistry: In chemistry, we often deal with incredibly large numbers of molecules or atoms in a given substance. Moles, a fundamental unit in chemistry, are frequently expressed in scientific notation. Avogadro's number, the number of molecules in one mole of a substance, is approximately 6.02 x 10<sup>23</sup>.
-
Biology: In biology, we might encounter vast numbers of microorganisms in a sample or very tiny measurements related to cellular structures. Scientific notation helps simplify the presentation and manipulation of this data It's one of those things that adds up. That alone is useful..
-
Computer Science: Data storage capacity is often represented using scientific notation, for example, terabytes (TB) or petabytes (PB). This allows for efficient communication of massive amounts of data And that's really what it comes down to..
-
Engineering: In various engineering disciplines, scientific notation has a big impact in representing large dimensions (like those involved in civil engineering projects) or very small tolerances in precision manufacturing Most people skip this — try not to..
Converting Numbers to and from Scientific Notation
Converting a number to scientific notation involves moving the decimal point until only one digit remains to the left of it, and then expressing the resulting number as a product of a coefficient and a power of 10. The exponent indicates how many places the decimal point was moved. A move to the left results in a positive exponent; a move to the right results in a negative exponent Simple as that..
-
Example (large number): Convert 3,450,000 to scientific notation. Move the decimal point six places to the left: 3.45 x 10<sup>6</sup>
-
Example (small number): Convert 0.0000027 to scientific notation. Move the decimal point six places to the right: 2.7 x 10<sup>-6</sup>
Converting from scientific notation to standard notation involves simply moving the decimal point according to the exponent. A positive exponent means moving the decimal point to the right; a negative exponent means moving it to the left.
Advanced Concepts and Applications
Beyond the basics, scientific notation finds applications in more advanced scientific and mathematical concepts:
-
Logarithms: Scientific notation is closely related to logarithms, which are essential tools in many scientific and engineering applications. Logarithms simplify calculations involving large ranges of values, especially in fields like acoustics and seismology.
-
Statistical Analysis: In statistics, handling large datasets often requires the use of scientific notation for clarity and efficiency in presenting and analyzing results And that's really what it comes down to. Practical, not theoretical..
-
Computational Physics and Chemistry: Simulations and modeling often involve extremely large or small numbers, and scientific notation is vital for handling the computational aspects of these analyses.
Frequently Asked Questions (FAQ)
Q1: What if the coefficient is not between 1 and 10?
A1: If the coefficient is not between 1 and 10, you need to adjust it. Take this: 25 x 10<sup>3</sup> should be rewritten as 2.5 x 10<sup>4</sup> And that's really what it comes down to..
Q2: How do I perform calculations with numbers in different units?
A2: Before performing calculations with numbers in different units, you need to convert them to the same unit. As an example, if you're working with kilometers and meters, convert them both to either kilometers or meters before adding or subtracting.
Q3: Can negative numbers be expressed in scientific notation?
A3: Yes, negative numbers can be expressed in scientific notation. Worth adding: the sign of the number is simply placed in front of the coefficient. Now, for example, -2. 5 x 10<sup>3</sup> Less friction, more output..
Q4: Is scientific notation only used in science?
A4: While predominantly used in science and engineering, scientific notation is a useful tool in any context involving very large or very small numbers. It promotes clarity and efficiency in handling such numbers Worth knowing..
Q5: Are there any limitations to scientific notation?
A5: While extremely useful, scientific notation doesn't provide a means for representing irrational numbers (like pi or the square root of 2) precisely. It offers an approximation for these numbers, suitable for many practical calculations but not representing their true, infinite nature That alone is useful..
Conclusion
The seemingly simple expression "1.5 x 10" opens the door to a vast world of numerical representation and manipulation. By mastering this fundamental concept, you equip yourself with a powerful tool for tackling complex scientific and mathematical problems, bridging the gap between abstract concepts and real-world applications. Understanding scientific notation is not just about manipulating numbers; it's about gaining a deeper appreciation for the scale of the universe, from the immensity of space to the intricacies of the atomic realm. The ability to understand and apply scientific notation is a cornerstone of scientific literacy and effective communication across diverse disciplines That's the whole idea..